Molecular diagnostics is the fastest-growing segment in the IVD market, driven by infectious diseases and oncology. This market is growing due an increasing pool in the aging population across the globe, a concurrent augmentation in the prevalence of chronic diseases, and the need for the diagnosis of such diseases at early stages. The market is driven by increasing awareness and acceptance of personalized medicines, advancements in molecular techniques, inherited disease testing, and prenatal testing. However, persistent reimbursement issues and delays in the approval of molecular diagnostic tests are expected to curb the growth of this market.
The Centers for Medicare & Medicaid Services (CMS) has estimated that healthcare spending in the U.S. is expected to grow from $2.7 trillion in 2011 to about $4.6 trillion in 2017, at a CAGR of 6.8% for the same period. Similarly, in emerging countries, awareness and an increasing middle-class population, with a more disposable income to spend on healthcare, will be a driving force for the growth of this market.
The report ‘European Molecular Diagnostics Market forecast for 2018’ analyzes the market by segments such as instruments and reagents.
Europe holds second largest market position. Germany commanded the largest share of 20% with $311.3 million in 2013, and is expected to reach $454.3 million by 2018, at a CAGR of 8.2%, for the given period.
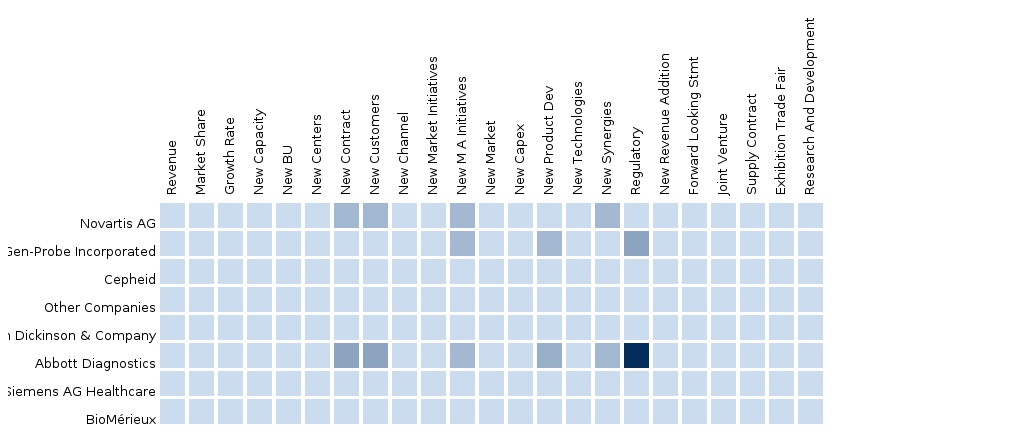
The report also provides an extensive competitive landscaping of companies operating in this market. The major players are Roche Diagnostics (Switzerland), Novartis (Switzerland), Gen-probe (U.S.), Qiagen (The Netherlands), and Abbott Diagnostics (U.S.). Segment and country-specific company shares, news & deals, mergers and acquisitions, segment specific pipeline products, product approvals, and product recalls of the major companies have been detailed.
Customization Options
Along with the market data, you can also customize MMM assessments that meet your company’s specific needs. Customize to get comprehensive industry standard and deep dive analysis of the following parameters:
Opportunity Analysis
- Unmet needs, revenue pockets, and potential areas for expansion
MDx Business Model
- Comprehensive review of key component providers, assay providers, sole service providers, and complete solution providers
Current and Emerging Products
- Analysis of current and emerging molecular diagnostics and immunodiagnostic tests
- Review of current instrumentation technologies
Pricing Trends
- Pricing trend of new high value* MDx tests
Product Analysis
- Usage pattern (in-depth trend analysis) of products (segment wise)
- Product matrix which gives a detailed comparison of product portfolio of each company mapped at country and sub-segment level
- End-user adoption rate analysis of the products (segment wise and country wise)
- Comprehensive coverage of product approvals, pipeline products, and product recalls
Brand/Product Perception Matrix
- Comprehensive study of customers perception and behavior through our inbuilt social connect tool checking the virility and tonality of blogs
- Analysis of overall brand usage and familiarity and brand advocacy distribution (detractor/neutral/familiar)
Alternative Products: Impact analysis
MMM’s healthcare decision making quadrant is an innovative and useful quadrant for vendors who wish to analyze the potential growth markets based on parameters like patient dynamics (patient pool, epidemiology of disease, preference towards surgeries/alternative therapies) and macroeconomic indicators (number of hospitals and orthopedic clinics, reimbursement scenario, diagnosis rate, treatment rate and healthcare expenditure).
1 Introduction
1.1 Objective of the study
1.2 Market Definitions
1.3 Market Segmentation & Aspects Covered
1.4 Research Methodology
1.4.1 Assumptions (Market Size, Forecast, etc)
2 Executive Summary
3 Market Overview
4 Molecular Diagnostics-Europe, by Applications
4.1 Split by Geography
4.1 Molecular Diagnostics-Spain by Applications
4.1 Molecular Diagnostics-United Kingdom by Applications
4.1 Molecular Diagnostics-France by Applications
4.1 Molecular Diagnostics-Germany by Applications
4.1 Molecular Diagnostics-Italy by Applications
4.1 Molecular Diagnostics-Rest of Europe by Applications
4.2 Molecular Diagnostics-Genetic Testing-Europe
4.2.1 Molecular Diagnostics-Genetic Testing-Europe, by Geographies
4.2.1.1 Molecular Diagnostics-Genetic Testing-Spain
4.2.1.2 Molecular Diagnostics-Genetic Testing-United Kingdom
4.2.1.3 Molecular Diagnostics-Genetic Testing-France
4.2.1.4 Molecular Diagnostics-Genetic Testing-Germany
4.2.1.5 Molecular Diagnostics-Genetic Testing-Italy
4.2.1.6 Molecular Diagnostics-Genetic Testing-Rest of Europe
4.3 Molecular Diagnostics-Microbiology-Europe
4.3.1 Molecular Diagnostics-Microbiology-Europe, by Geographies
4.3.1.1 Molecular Diagnostics-Microbiology-Spain
4.3.1.2 Molecular Diagnostics-Microbiology-United Kingdom
4.3.1.3 Molecular Diagnostics-Microbiology-France
4.3.1.4 Molecular Diagnostics-Microbiology-Germany
4.3.1.5 Molecular Diagnostics-Microbiology-Italy
4.3.1.6 Molecular Diagnostics-Microbiology-Rest of Europe
4.4 Molecular Diagnostics-Chlamydia trachomatis/ Neisseria gonorrhoeae-Europe
4.4.1 Molecular Diagnostics-Chlamydia trachomatis/ Neisseria gonorrhoeae-Europe, by Geographies
4.4.1.1 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.2 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.3 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.4 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.5 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.6 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.7 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.8 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.9 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.10 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.11 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.12 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.13 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.14 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.15 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.16 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.17 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.4.1.18 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
4.5 Molecular Diagnostics-Hepatitis B-Europe
4.5.1 Molecular Diagnostics-Hepatitis B-Europe, by Geographies
4.5.1.1 Molecular Diagnostics-Spain-Hepatitis B
4.5.1.2 Molecular Diagnostics-Spain-Hepatitis B
4.5.1.3 Molecular Diagnostics-Spain-Hepatitis B
4.5.1.4 Molecular Diagnostics-United Kingdom-Hepatitis B
4.5.1.5 Molecular Diagnostics-United Kingdom-Hepatitis B
4.5.1.6 Molecular Diagnostics-United Kingdom-Hepatitis B
4.5.1.7 Molecular Diagnostics-France-Hepatitis B
4.5.1.8 Molecular Diagnostics-France-Hepatitis B
4.5.1.9 Molecular Diagnostics-France-Hepatitis B
4.5.1.10 Molecular Diagnostics-Germany-Hepatitis B
4.5.1.11 Molecular Diagnostics-Germany-Hepatitis B
4.5.1.12 Molecular Diagnostics-Germany-Hepatitis B
4.5.1.13 Molecular Diagnostics-Italy-Hepatitis B
4.5.1.14 Molecular Diagnostics-Italy-Hepatitis B
4.5.1.15 Molecular Diagnostics-Italy-Hepatitis B
4.5.1.16 Molecular Diagnostics-Rest of Europe-Hepatitis B
4.5.1.17 Molecular Diagnostics-Rest of Europe-Hepatitis B
4.5.1.18 Molecular Diagnostics-Rest of Europe-Hepatitis B
4.6 Molecular Diagnostics--Europe
4.6.1 Molecular Diagnostics--Europe, by Geographies
4.6.1.1 Molecular Diagnostics--Spain
4.6.1.2 Molecular Diagnostics--United Kingdom
4.6.1.3 Molecular Diagnostics--France
4.6.1.4 Molecular Diagnostics--Germany
4.6.1.5 Molecular Diagnostics--Italy
4.6.1.6 Molecular Diagnostics--Rest of Europe
4.7 Molecular Diagnostics-Breast Cancer-Europe
4.7.1 Molecular Diagnostics-Breast Cancer-Europe, by Geographies
4.7.1.1 Molecular Diagnostics-Spain-Breast Cancer
4.7.1.2 Molecular Diagnostics-Spain-Breast Cancer
4.7.1.3 Molecular Diagnostics-Spain-Breast Cancer
4.7.1.4 Molecular Diagnostics-United Kingdom-Breast Cancer
4.7.1.5 Molecular Diagnostics-United Kingdom-Breast Cancer
4.7.1.6 Molecular Diagnostics-United Kingdom-Breast Cancer
4.7.1.7 Molecular Diagnostics-France-Breast Cancer
4.7.1.8 Molecular Diagnostics-France-Breast Cancer
4.7.1.9 Molecular Diagnostics-France-Breast Cancer
4.7.1.10 Molecular Diagnostics-Germany-Breast Cancer
4.7.1.11 Molecular Diagnostics-Germany-Breast Cancer
4.7.1.12 Molecular Diagnostics-Germany-Breast Cancer
4.7.1.13 Molecular Diagnostics-Italy-Breast Cancer
4.7.1.14 Molecular Diagnostics-Italy-Breast Cancer
4.7.1.15 Molecular Diagnostics-Italy-Breast Cancer
4.7.1.16 Molecular Diagnostics-Rest of Europe-Breast Cancer
4.7.1.17 Molecular Diagnostics-Rest of Europe-Breast Cancer
4.7.1.18 Molecular Diagnostics-Rest of Europe-Breast Cancer
4.8 Molecular Diagnostics-Human Papillomavirus-Europe
4.8.1 Molecular Diagnostics-Human Papillomavirus-Europe, by Geographies
4.8.1.1 Molecular Diagnostics-Spain-Human Papillomavirus
4.8.1.2 Molecular Diagnostics-Spain-Human Papillomavirus
4.8.1.3 Molecular Diagnostics-Spain-Human Papillomavirus
4.8.1.4 Molecular Diagnostics-United Kingdom-Human Papillomavirus
4.8.1.5 Molecular Diagnostics-United Kingdom-Human Papillomavirus
4.8.1.6 Molecular Diagnostics-United Kingdom-Human Papillomavirus
4.8.1.7 Molecular Diagnostics-France-Human Papillomavirus
4.8.1.8 Molecular Diagnostics-France-Human Papillomavirus
4.8.1.9 Molecular Diagnostics-France-Human Papillomavirus
4.8.1.10 Molecular Diagnostics-Germany-Human Papillomavirus
4.8.1.11 Molecular Diagnostics-Germany-Human Papillomavirus
4.8.1.12 Molecular Diagnostics-Germany-Human Papillomavirus
4.8.1.13 Molecular Diagnostics-Italy-Human Papillomavirus
4.8.1.14 Molecular Diagnostics-Italy-Human Papillomavirus
4.8.1.15 Molecular Diagnostics-Italy-Human Papillomavirus
4.8.1.16 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
4.8.1.17 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
4.8.1.18 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
4.9 Molecular Diagnostics-HIV/AIDS-Europe
4.9.1 Molecular Diagnostics-HIV/AIDS-Europe, by Geographies
4.9.1.1 Molecular Diagnostics-Spain-HIV/AIDS
4.9.1.2 Molecular Diagnostics-Spain-HIV/AIDS
4.9.1.3 Molecular Diagnostics-Spain-HIV/AIDS
4.9.1.4 Molecular Diagnostics-United Kingdom-HIV/AIDS
4.9.1.5 Molecular Diagnostics-United Kingdom-HIV/AIDS
4.9.1.6 Molecular Diagnostics-United Kingdom-HIV/AIDS
4.9.1.7 Molecular Diagnostics-France-HIV/AIDS
4.9.1.8 Molecular Diagnostics-France-HIV/AIDS
4.9.1.9 Molecular Diagnostics-France-HIV/AIDS
4.9.1.10 Molecular Diagnostics-Germany-HIV/AIDS
4.9.1.11 Molecular Diagnostics-Germany-HIV/AIDS
4.9.1.12 Molecular Diagnostics-Germany-HIV/AIDS
4.9.1.13 Molecular Diagnostics-Italy-HIV/AIDS
4.9.1.14 Molecular Diagnostics-Italy-HIV/AIDS
4.9.1.15 Molecular Diagnostics-Italy-HIV/AIDS
4.9.1.16 Molecular Diagnostics-Rest of Europe-HIV/AIDS
4.9.1.17 Molecular Diagnostics-Rest of Europe-HIV/AIDS
4.9.1.18 Molecular Diagnostics-Rest of Europe-HIV/AIDS
4.10 Molecular Diagnostics-Tuberculosis-Europe
4.10.1 Molecular Diagnostics-Tuberculosis-Europe, by Geographies
4.10.1.1 Molecular Diagnostics-Spain-Tuberculosis
4.10.1.2 Molecular Diagnostics-Spain-Tuberculosis
4.10.1.3 Molecular Diagnostics-Spain-Tuberculosis
4.10.1.4 Molecular Diagnostics-United Kingdom-Tuberculosis
4.10.1.5 Molecular Diagnostics-United Kingdom-Tuberculosis
4.10.1.6 Molecular Diagnostics-United Kingdom-Tuberculosis
4.10.1.7 Molecular Diagnostics-France-Tuberculosis
4.10.1.8 Molecular Diagnostics-France-Tuberculosis
4.10.1.9 Molecular Diagnostics-France-Tuberculosis
4.10.1.10 Molecular Diagnostics-Germany-Tuberculosis
4.10.1.11 Molecular Diagnostics-Germany-Tuberculosis
4.10.1.12 Molecular Diagnostics-Germany-Tuberculosis
4.10.1.13 Molecular Diagnostics-Italy-Tuberculosis
4.10.1.14 Molecular Diagnostics-Italy-Tuberculosis
4.10.1.15 Molecular Diagnostics-Italy-Tuberculosis
4.10.1.16 Molecular Diagnostics-Rest of Europe-Tuberculosis
4.10.1.17 Molecular Diagnostics-Rest of Europe-Tuberculosis
4.10.1.18 Molecular Diagnostics-Rest of Europe-Tuberculosis
4.11 Molecular Diagnostics-Hepatitis C-Europe
4.11.1 Molecular Diagnostics-Hepatitis C-Europe, by Geographies
4.11.1.1 Molecular Diagnostics-Spain-Hepatitis C
4.11.1.2 Molecular Diagnostics-Spain-Hepatitis C
4.11.1.3 Molecular Diagnostics-Spain-Hepatitis C
4.11.1.4 Molecular Diagnostics-United Kingdom-Hepatitis C
4.11.1.5 Molecular Diagnostics-United Kingdom-Hepatitis C
4.11.1.6 Molecular Diagnostics-United Kingdom-Hepatitis C
4.11.1.7 Molecular Diagnostics-France-Hepatitis C
4.11.1.8 Molecular Diagnostics-France-Hepatitis C
4.11.1.9 Molecular Diagnostics-France-Hepatitis C
4.11.1.10 Molecular Diagnostics-Germany-Hepatitis C
4.11.1.11 Molecular Diagnostics-Germany-Hepatitis C
4.11.1.12 Molecular Diagnostics-Germany-Hepatitis C
4.11.1.13 Molecular Diagnostics-Italy-Hepatitis C
4.11.1.14 Molecular Diagnostics-Italy-Hepatitis C
4.11.1.15 Molecular Diagnostics-Italy-Hepatitis C
4.11.1.16 Molecular Diagnostics-Rest of Europe-Hepatitis C
4.11.1.17 Molecular Diagnostics-Rest of Europe-Hepatitis C
4.11.1.18 Molecular Diagnostics-Rest of Europe-Hepatitis C
4.12 Molecular Diagnostics-Methicillin-resistant Staphylococcus aureus (MRSA)-Europe
4.12.1 Molecular Diagnostics-Methicillin-resistant Staphylococcus aureus (MRSA)-Europe, by Geographies
4.12.1.1 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.2 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.3 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.4 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.5 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.6 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.7 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.8 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.9 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.10 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.11 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.12 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.13 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.14 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.15 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.16 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.17 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
4.12.1.18 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
4.13 Molecular Diagnostics-Colorectal Cancer-Europe
4.13.1 Molecular Diagnostics-Colorectal Cancer-Europe, by Geographies
4.13.1.1 Molecular Diagnostics-Spain-Colorectal Cancer
4.13.1.2 Molecular Diagnostics-Spain-Colorectal Cancer
4.13.1.3 Molecular Diagnostics-Spain-Colorectal Cancer
4.13.1.4 Molecular Diagnostics-United Kingdom-Colorectal Cancer
4.13.1.5 Molecular Diagnostics-United Kingdom-Colorectal Cancer
4.13.1.6 Molecular Diagnostics-United Kingdom-Colorectal Cancer
4.13.1.7 Molecular Diagnostics-France-Colorectal Cancer
4.13.1.8 Molecular Diagnostics-France-Colorectal Cancer
4.13.1.9 Molecular Diagnostics-France-Colorectal Cancer
4.13.1.10 Molecular Diagnostics-Germany-Colorectal Cancer
4.13.1.11 Molecular Diagnostics-Germany-Colorectal Cancer
4.13.1.12 Molecular Diagnostics-Germany-Colorectal Cancer
4.13.1.13 Molecular Diagnostics-Italy-Colorectal Cancer
4.13.1.14 Molecular Diagnostics-Italy-Colorectal Cancer
4.13.1.15 Molecular Diagnostics-Italy-Colorectal Cancer
4.13.1.16 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
4.13.1.17 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
4.13.1.18 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
4.14 Molecular Diagnostics-Prostate Cancer-Europe
4.14.1 Molecular Diagnostics-Prostate Cancer-Europe, by Geographies
4.14.1.1 Molecular Diagnostics-Spain-Prostate Cancer
4.14.1.2 Molecular Diagnostics-Spain-Prostate Cancer
4.14.1.3 Molecular Diagnostics-Spain-Prostate Cancer
4.14.1.4 Molecular Diagnostics-United Kingdom-Prostate Cancer
4.14.1.5 Molecular Diagnostics-United Kingdom-Prostate Cancer
4.14.1.6 Molecular Diagnostics-United Kingdom-Prostate Cancer
4.14.1.7 Molecular Diagnostics-France-Prostate Cancer
4.14.1.8 Molecular Diagnostics-France-Prostate Cancer
4.14.1.9 Molecular Diagnostics-France-Prostate Cancer
4.14.1.10 Molecular Diagnostics-Germany-Prostate Cancer
4.14.1.11 Molecular Diagnostics-Germany-Prostate Cancer
4.14.1.12 Molecular Diagnostics-Germany-Prostate Cancer
4.14.1.13 Molecular Diagnostics-Italy-Prostate Cancer
4.14.1.14 Molecular Diagnostics-Italy-Prostate Cancer
4.14.1.15 Molecular Diagnostics-Italy-Prostate Cancer
4.14.1.16 Molecular Diagnostics-Rest of Europe-Prostate Cancer
4.14.1.17 Molecular Diagnostics-Rest of Europe-Prostate Cancer
4.14.1.18 Molecular Diagnostics-Rest of Europe-Prostate Cancer
5 Molecular Diagnostics-Europe, by Endusers
5.1 Split by Geography
5.2 Molecular Diagnostics-Spain by Endusers
5.1 Molecular Diagnostics-United Kingdom by Endusers
5.1 Molecular Diagnostics-France by Endusers
5.1 Molecular Diagnostics-Germany by Endusers
5.1 Molecular Diagnostics-Italy by Endusers
5.1 Molecular Diagnostics-Rest of Europe by Endusers
5.2 Molecular Diagnostics-Europe-Hospitals
5.2.1 Molecular Diagnostics-Europe-Hospitals, by Geographies
5.2.1.1 Molecular Diagnostics-Spain-Hospitals
5.2.1.2 Molecular Diagnostics-United Kingdom-Hospitals
5.2.1.3 Molecular Diagnostics-France-Hospitals
5.2.1.4 Molecular Diagnostics-Germany-Hospitals
5.2.1.5 Molecular Diagnostics-Italy-Hospitals
5.2.1.6 Molecular Diagnostics-Rest of Europe-Hospitals
5.3 Molecular Diagnostics-Europe-Medical & Diagnostic Laboratories
5.3.1 Molecular Diagnostics-Europe-Medical & Diagnostic Laboratories, by Geographies
5.3.1.1 Molecular Diagnostics-Spain-Medical & Diagnostic Laboratories
5.3.1.2 Molecular Diagnostics-United Kingdom-Medical & Diagnostic Laboratories
5.3.1.3 Molecular Diagnostics-France-Medical & Diagnostic Laboratories
5.3.1.4 Molecular Diagnostics-Germany-Medical & Diagnostic Laboratories
5.3.1.5 Molecular Diagnostics-Italy-Medical & Diagnostic Laboratories
5.3.1.6 Molecular Diagnostics-Rest of Europe-Medical & Diagnostic Laboratories
5.4 Molecular Diagnostics--Europe
5.4.1 Molecular Diagnostics--Europe, by Geographies
5.4.1.1 Molecular Diagnostics--Spain
5.4.1.2 Molecular Diagnostics--United Kingdom
5.4.1.3 Molecular Diagnostics--France
5.4.1.4 Molecular Diagnostics--Germany
5.4.1.5 Molecular Diagnostics--Italy
5.4.1.6 Molecular Diagnostics--Rest of Europe
6 Molecular Diagnostics-Europe, by Technologies
6.1 Split by Geography
6.3 Molecular Diagnostics-Spain by Technologies
6.1 Molecular Diagnostics-United Kingdom by Technologies
6.1 Molecular Diagnostics-France by Technologies
6.1 Molecular Diagnostics-Germany by Technologies
6.1 Molecular Diagnostics-Italy by Technologies
6.1 Molecular Diagnostics-Rest of Europe by Technologies
6.2 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Europe
6.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Europe, by Geographies
6.2.1.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Spain
6.2.1.2 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-United Kingdom
6.2.1.3 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-France
6.2.1.4 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Germany
6.2.1.5 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Italy
6.2.1.6 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Rest of Europe
6.3 Molecular Diagnostics--Europe
6.3.1 Molecular Diagnostics--Europe, by Geographies
6.3.1.1 Molecular Diagnostics--Spain
6.3.1.2 Molecular Diagnostics--United Kingdom
6.3.1.3 Molecular Diagnostics--France
6.3.1.4 Molecular Diagnostics--Germany
6.3.1.5 Molecular Diagnostics--Italy
6.3.1.6 Molecular Diagnostics--Rest of Europe
7 Molecular Diagnostics-Europe, by Products
7.1 Split by Geography
7.4 Molecular Diagnostics-Spain by Products
7.1 Molecular Diagnostics-United Kingdom by Products
7.1 Molecular Diagnostics-France by Products
7.1 Molecular Diagnostics-Germany by Products
7.1 Molecular Diagnostics-Italy by Products
7.1 Molecular Diagnostics-Rest of Europe by Products
7.2 Molecular Diagnostics Instruments-Europe
7.2.1 Molecular Diagnostics Instruments-Europe, by Geographies
7.2.1.1 Molecular Diagnostics Instruments-Spain
7.2.1.2 Molecular Diagnostics Instruments-United Kingdom
7.2.1.3 Molecular Diagnostics Instruments-France
7.2.1.4 Molecular Diagnostics Instruments-Germany
7.2.1.5 Molecular Diagnostics Instruments-Italy
7.2.1.6 Molecular Diagnostics Instruments-Rest of Europe
7.3 Molecular Diagnostics Reagents-Europe
7.3.1 Molecular Diagnostics Reagents-Europe, by Geographies
7.3.1.1 Molecular Diagnostics Reagents-Spain
7.3.1.2 Molecular Diagnostics Reagents-United Kingdom
7.3.1.3 Molecular Diagnostics Reagents-France
7.3.1.4 Molecular Diagnostics Reagents-Germany
7.3.1.5 Molecular Diagnostics Reagents-Italy
7.3.1.6 Molecular Diagnostics Reagents-Rest of Europe
7.4 Molecular Diagnostics Services-Europe
7.4.1 Molecular Diagnostics Services-Europe, by Geographies
7.4.1.1 Molecular Diagnostics Services-Spain
7.4.1.2 Molecular Diagnostics Services-United Kingdom
7.4.1.3 Molecular Diagnostics Services-France
7.4.1.4 Molecular Diagnostics Services-Germany
7.4.1.5 Molecular Diagnostics Services-Italy
7.4.1.6 Molecular Diagnostics Services-Rest of Europe
8 Molecular Diagnostics-Europe, by Geographies
8.1 Molecular Diagnostics-Spain
8.1.1 Molecular Diagnostics-Spain, by Endusers
8.1.1.1 Molecular Diagnostics-Spain-Hospitals
8.1.1.2 Molecular Diagnostics-Spain-Medical & Diagnostic Laboratories
8.1.1.3 Molecular Diagnostics--Spain
8.1.2 Molecular Diagnostics-Spain, by Technologies
8.1.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Spain
8.1.2.2 Molecular Diagnostics--Spain
8.1.3 Molecular Diagnostics-Spain, by Applications
8.1.3.1 Molecular Diagnostics-Microbiology-Spain
8.1.3.2 Molecular Diagnostics--Spain
8.1.3.3 Molecular Diagnostics-Genetic Testing-Spain
8.1.3.4 Molecular Diagnostics-Spain-Hepatitis B
8.1.3.5 Molecular Diagnostics-Spain-Hepatitis B
8.1.3.6 Molecular Diagnostics-Spain-Hepatitis B
8.1.3.7 Molecular Diagnostics-Spain-Hepatitis C
8.1.3.8 Molecular Diagnostics-Spain-HIV/AIDS
8.1.3.9 Molecular Diagnostics-Spain-Human Papillomavirus
8.1.3.10 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
8.1.3.11 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.1.3.12 Molecular Diagnostics-Spain-Tuberculosis
8.1.3.13 Molecular Diagnostics-Spain-Prostate Cancer
8.1.3.14 Molecular Diagnostics-Spain-Breast Cancer
8.1.3.15 Molecular Diagnostics-Spain-Colorectal Cancer
8.1.3.16 Molecular Diagnostics-Spain-Hepatitis C
8.1.3.17 Molecular Diagnostics-Spain-HIV/AIDS
8.1.3.18 Molecular Diagnostics-Spain-Human Papillomavirus
8.1.3.19 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
8.1.3.20 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.1.3.21 Molecular Diagnostics-Spain-Tuberculosis
8.1.3.22 Molecular Diagnostics-Spain-Prostate Cancer
8.1.3.23 Molecular Diagnostics-Spain-Breast Cancer
8.1.3.24 Molecular Diagnostics-Spain-Colorectal Cancer
8.1.3.25 Molecular Diagnostics-Spain-Hepatitis C
8.1.3.26 Molecular Diagnostics-Spain-HIV/AIDS
8.1.3.27 Molecular Diagnostics-Spain-Human Papillomavirus
8.1.3.28 Molecular Diagnostics-Spain-Methicillin-resistant Staphylococcus aureus (MRSA)
8.1.3.29 Molecular Diagnostics-Spain-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.1.3.30 Molecular Diagnostics-Spain-Tuberculosis
8.1.3.31 Molecular Diagnostics-Spain-Prostate Cancer
8.1.3.32 Molecular Diagnostics-Spain-Breast Cancer
8.1.3.33 Molecular Diagnostics-Spain-Colorectal Cancer
8.1.4 Molecular Diagnostics-Spain, by Products
8.1.4.1 Molecular Diagnostics Instruments-Spain
8.1.4.2 Molecular Diagnostics Reagents-Spain
8.1.4.3 Molecular Diagnostics Services-Spain
8.2 Molecular Diagnostics-United Kingdom
8.2.1 Molecular Diagnostics-United Kingdom, by Endusers
8.2.1.1 Molecular Diagnostics-United Kingdom-Hospitals
8.2.1.2 Molecular Diagnostics-United Kingdom-Medical & Diagnostic Laboratories
8.2.1.3 Molecular Diagnostics--United Kingdom
8.2.2 Molecular Diagnostics-United Kingdom, by Technologies
8.2.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-United Kingdom
8.2.2.2 Molecular Diagnostics--United Kingdom
8.2.3 Molecular Diagnostics-United Kingdom, by Applications
8.2.3.1 Molecular Diagnostics-Microbiology-United Kingdom
8.2.3.2 Molecular Diagnostics--United Kingdom
8.2.3.3 Molecular Diagnostics-Genetic Testing-United Kingdom
8.2.3.4 Molecular Diagnostics-United Kingdom-Hepatitis B
8.2.3.5 Molecular Diagnostics-United Kingdom-Hepatitis C
8.2.3.6 Molecular Diagnostics-United Kingdom-HIV/AIDS
8.2.3.7 Molecular Diagnostics-United Kingdom-Human Papillomavirus
8.2.3.8 Molecular Diagnostics-United Kingdom-Hepatitis B
8.2.3.9 Molecular Diagnostics-United Kingdom-Hepatitis C
8.2.3.10 Molecular Diagnostics-United Kingdom-HIV/AIDS
8.2.3.11 Molecular Diagnostics-United Kingdom-Human Papillomavirus
8.2.3.12 Molecular Diagnostics-United Kingdom-Hepatitis B
8.2.3.13 Molecular Diagnostics-United Kingdom-Hepatitis C
8.2.3.14 Molecular Diagnostics-United Kingdom-HIV/AIDS
8.2.3.15 Molecular Diagnostics-United Kingdom-Human Papillomavirus
8.2.3.16 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
8.2.3.17 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.2.3.18 Molecular Diagnostics-United Kingdom-Tuberculosis
8.2.3.19 Molecular Diagnostics-United Kingdom-Prostate Cancer
8.2.3.20 Molecular Diagnostics-United Kingdom-Breast Cancer
8.2.3.21 Molecular Diagnostics-United Kingdom-Colorectal Cancer
8.2.3.22 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
8.2.3.23 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.2.3.24 Molecular Diagnostics-United Kingdom-Tuberculosis
8.2.3.25 Molecular Diagnostics-United Kingdom-Prostate Cancer
8.2.3.26 Molecular Diagnostics-United Kingdom-Breast Cancer
8.2.3.27 Molecular Diagnostics-United Kingdom-Colorectal Cancer
8.2.3.28 Molecular Diagnostics-United Kingdom-Methicillin-resistant Staphylococcus aureus (MRSA)
8.2.3.29 Molecular Diagnostics-United Kingdom-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.2.3.30 Molecular Diagnostics-United Kingdom-Tuberculosis
8.2.3.31 Molecular Diagnostics-United Kingdom-Prostate Cancer
8.2.3.32 Molecular Diagnostics-United Kingdom-Breast Cancer
8.2.3.33 Molecular Diagnostics-United Kingdom-Colorectal Cancer
8.2.4 Molecular Diagnostics-United Kingdom, by Products
8.2.4.1 Molecular Diagnostics Instruments-United Kingdom
8.2.4.2 Molecular Diagnostics Reagents-United Kingdom
8.2.4.3 Molecular Diagnostics Services-United Kingdom
8.3 Molecular Diagnostics-France
8.3.1 Molecular Diagnostics-France, by Endusers
8.3.1.1 Molecular Diagnostics-France-Hospitals
8.3.1.2 Molecular Diagnostics-France-Medical & Diagnostic Laboratories
8.3.1.3 Molecular Diagnostics--France
8.3.2 Molecular Diagnostics-France, by Technologies
8.3.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-France
8.3.2.2 Molecular Diagnostics--France
8.3.3 Molecular Diagnostics-France, by Applications
8.3.3.1 Molecular Diagnostics-Microbiology-France
8.3.3.2 Molecular Diagnostics--France
8.3.3.3 Molecular Diagnostics-Genetic Testing-France
8.3.3.4 Molecular Diagnostics-France-Hepatitis B
8.3.3.5 Molecular Diagnostics-France-Hepatitis C
8.3.3.6 Molecular Diagnostics-France-HIV/AIDS
8.3.3.7 Molecular Diagnostics-France-Human Papillomavirus
8.3.3.8 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
8.3.3.9 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.3.3.10 Molecular Diagnostics-France-Hepatitis B
8.3.3.11 Molecular Diagnostics-France-Hepatitis C
8.3.3.12 Molecular Diagnostics-France-HIV/AIDS
8.3.3.13 Molecular Diagnostics-France-Human Papillomavirus
8.3.3.14 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
8.3.3.15 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.3.3.16 Molecular Diagnostics-France-Hepatitis B
8.3.3.17 Molecular Diagnostics-France-Hepatitis C
8.3.3.18 Molecular Diagnostics-France-HIV/AIDS
8.3.3.19 Molecular Diagnostics-France-Human Papillomavirus
8.3.3.20 Molecular Diagnostics-France-Methicillin-resistant Staphylococcus aureus (MRSA)
8.3.3.21 Molecular Diagnostics-France-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.3.3.22 Molecular Diagnostics-France-Tuberculosis
8.3.3.23 Molecular Diagnostics-France-Prostate Cancer
8.3.3.24 Molecular Diagnostics-France-Breast Cancer
8.3.3.25 Molecular Diagnostics-France-Colorectal Cancer
8.3.3.26 Molecular Diagnostics-France-Tuberculosis
8.3.3.27 Molecular Diagnostics-France-Prostate Cancer
8.3.3.28 Molecular Diagnostics-France-Breast Cancer
8.3.3.29 Molecular Diagnostics-France-Colorectal Cancer
8.3.3.30 Molecular Diagnostics-France-Tuberculosis
8.3.3.31 Molecular Diagnostics-France-Prostate Cancer
8.3.3.32 Molecular Diagnostics-France-Breast Cancer
8.3.3.33 Molecular Diagnostics-France-Colorectal Cancer
8.3.4 Molecular Diagnostics-France, by Products
8.3.4.1 Molecular Diagnostics Instruments-France
8.3.4.2 Molecular Diagnostics Reagents-France
8.3.4.3 Molecular Diagnostics Services-France
8.4 Molecular Diagnostics-Germany
8.4.1 Molecular Diagnostics-Germany, by Endusers
8.4.1.1 Molecular Diagnostics-Germany-Hospitals
8.4.1.2 Molecular Diagnostics-Germany-Medical & Diagnostic Laboratories
8.4.1.3 Molecular Diagnostics--Germany
8.4.2 Molecular Diagnostics-Germany, by Technologies
8.4.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Germany
8.4.2.2 Molecular Diagnostics--Germany
8.4.3 Molecular Diagnostics-Germany, by Applications
8.4.3.1 Molecular Diagnostics-Microbiology-Germany
8.4.3.2 Molecular Diagnostics--Germany
8.4.3.3 Molecular Diagnostics-Genetic Testing-Germany
8.4.3.4 Molecular Diagnostics-Germany-Hepatitis B
8.4.3.5 Molecular Diagnostics-Germany-Hepatitis C
8.4.3.6 Molecular Diagnostics-Germany-HIV/AIDS
8.4.3.7 Molecular Diagnostics-Germany-Human Papillomavirus
8.4.3.8 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
8.4.3.9 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.4.3.10 Molecular Diagnostics-Germany-Tuberculosis
8.4.3.11 Molecular Diagnostics-Germany-Prostate Cancer
8.4.3.12 Molecular Diagnostics-Germany-Breast Cancer
8.4.3.13 Molecular Diagnostics-Germany-Colorectal Cancer
8.4.3.14 Molecular Diagnostics-Germany-Hepatitis B
8.4.3.15 Molecular Diagnostics-Germany-Hepatitis C
8.4.3.16 Molecular Diagnostics-Germany-HIV/AIDS
8.4.3.17 Molecular Diagnostics-Germany-Human Papillomavirus
8.4.3.18 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
8.4.3.19 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.4.3.20 Molecular Diagnostics-Germany-Tuberculosis
8.4.3.21 Molecular Diagnostics-Germany-Prostate Cancer
8.4.3.22 Molecular Diagnostics-Germany-Breast Cancer
8.4.3.23 Molecular Diagnostics-Germany-Colorectal Cancer
8.4.3.24 Molecular Diagnostics-Germany-Hepatitis B
8.4.3.25 Molecular Diagnostics-Germany-Hepatitis C
8.4.3.26 Molecular Diagnostics-Germany-HIV/AIDS
8.4.3.27 Molecular Diagnostics-Germany-Human Papillomavirus
8.4.3.28 Molecular Diagnostics-Germany-Methicillin-resistant Staphylococcus aureus (MRSA)
8.4.3.29 Molecular Diagnostics-Germany-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.4.3.30 Molecular Diagnostics-Germany-Tuberculosis
8.4.3.31 Molecular Diagnostics-Germany-Prostate Cancer
8.4.3.32 Molecular Diagnostics-Germany-Breast Cancer
8.4.3.33 Molecular Diagnostics-Germany-Colorectal Cancer
8.4.4 Molecular Diagnostics-Germany, by Products
8.4.4.1 Molecular Diagnostics Instruments-Germany
8.4.4.2 Molecular Diagnostics Reagents-Germany
8.4.4.3 Molecular Diagnostics Services-Germany
8.5 Molecular Diagnostics-Italy
8.5.1 Molecular Diagnostics-Italy, by Endusers
8.5.1.1 Molecular Diagnostics-Italy-Hospitals
8.5.1.2 Molecular Diagnostics-Italy-Medical & Diagnostic Laboratories
8.5.1.3 Molecular Diagnostics--Italy
8.5.2 Molecular Diagnostics-Italy, by Technologies
8.5.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Italy
8.5.2.2 Molecular Diagnostics--Italy
8.5.3 Molecular Diagnostics-Italy, by Applications
8.5.3.1 Molecular Diagnostics-Microbiology-Italy
8.5.3.2 Molecular Diagnostics--Italy
8.5.3.3 Molecular Diagnostics-Genetic Testing-Italy
8.5.3.4 Molecular Diagnostics-Italy-Hepatitis B
8.5.3.5 Molecular Diagnostics-Italy-Hepatitis B
8.5.3.6 Molecular Diagnostics-Italy-Hepatitis B
8.5.3.7 Molecular Diagnostics-Italy-Hepatitis C
8.5.3.8 Molecular Diagnostics-Italy-HIV/AIDS
8.5.3.9 Molecular Diagnostics-Italy-Human Papillomavirus
8.5.3.10 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
8.5.3.11 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.5.3.12 Molecular Diagnostics-Italy-Tuberculosis
8.5.3.13 Molecular Diagnostics-Italy-Prostate Cancer
8.5.3.14 Molecular Diagnostics-Italy-Breast Cancer
8.5.3.15 Molecular Diagnostics-Italy-Colorectal Cancer
8.5.3.16 Molecular Diagnostics-Italy-Hepatitis C
8.5.3.17 Molecular Diagnostics-Italy-HIV/AIDS
8.5.3.18 Molecular Diagnostics-Italy-Human Papillomavirus
8.5.3.19 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
8.5.3.20 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.5.3.21 Molecular Diagnostics-Italy-Tuberculosis
8.5.3.22 Molecular Diagnostics-Italy-Prostate Cancer
8.5.3.23 Molecular Diagnostics-Italy-Breast Cancer
8.5.3.24 Molecular Diagnostics-Italy-Colorectal Cancer
8.5.3.25 Molecular Diagnostics-Italy-Hepatitis C
8.5.3.26 Molecular Diagnostics-Italy-HIV/AIDS
8.5.3.27 Molecular Diagnostics-Italy-Human Papillomavirus
8.5.3.28 Molecular Diagnostics-Italy-Methicillin-resistant Staphylococcus aureus (MRSA)
8.5.3.29 Molecular Diagnostics-Italy-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.5.3.30 Molecular Diagnostics-Italy-Tuberculosis
8.5.3.31 Molecular Diagnostics-Italy-Prostate Cancer
8.5.3.32 Molecular Diagnostics-Italy-Breast Cancer
8.5.3.33 Molecular Diagnostics-Italy-Colorectal Cancer
8.5.4 Molecular Diagnostics-Italy, by Products
8.5.4.1 Molecular Diagnostics Instruments-Italy
8.5.4.2 Molecular Diagnostics Reagents-Italy
8.5.4.3 Molecular Diagnostics Services-Italy
8.6 Molecular Diagnostics-Rest of Europe
8.6.1 Molecular Diagnostics-Rest of Europe, by Endusers
8.6.1.1 Molecular Diagnostics-Rest of Europe-Hospitals
8.6.1.2 Molecular Diagnostics-Rest of Europe-Medical & Diagnostic Laboratories
8.6.1.3 Molecular Diagnostics--Rest of Europe
8.6.2 Molecular Diagnostics-Rest of Europe, by Technologies
8.6.2.1 Molecular Diagnostics-Transcription-mediated Amplification (TMA)-Rest of Europe
8.6.2.2 Molecular Diagnostics--Rest of Europe
8.6.3 Molecular Diagnostics-Rest of Europe, by Applications
8.6.3.1 Molecular Diagnostics-Microbiology-Rest of Europe
8.6.3.2 Molecular Diagnostics--Rest of Europe
8.6.3.3 Molecular Diagnostics-Genetic Testing-Rest of Europe
8.6.3.4 Molecular Diagnostics-Rest of Europe-Hepatitis B
8.6.3.5 Molecular Diagnostics-Rest of Europe-Hepatitis C
8.6.3.6 Molecular Diagnostics-Rest of Europe-HIV/AIDS
8.6.3.7 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
8.6.3.8 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
8.6.3.9 Molecular Diagnostics-Rest of Europe-Hepatitis B
8.6.3.10 Molecular Diagnostics-Rest of Europe-Hepatitis C
8.6.3.11 Molecular Diagnostics-Rest of Europe-HIV/AIDS
8.6.3.12 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
8.6.3.13 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
8.6.3.14 Molecular Diagnostics-Rest of Europe-Hepatitis B
8.6.3.15 Molecular Diagnostics-Rest of Europe-Hepatitis C
8.6.3.16 Molecular Diagnostics-Rest of Europe-HIV/AIDS
8.6.3.17 Molecular Diagnostics-Rest of Europe-Human Papillomavirus
8.6.3.18 Molecular Diagnostics-Rest of Europe-Methicillin-resistant Staphylococcus aureus (MRSA)
8.6.3.19 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.6.3.20 Molecular Diagnostics-Rest of Europe-Tuberculosis
8.6.3.21 Molecular Diagnostics-Rest of Europe-Prostate Cancer
8.6.3.22 Molecular Diagnostics-Rest of Europe-Breast Cancer
8.6.3.23 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
8.6.3.24 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.6.3.25 Molecular Diagnostics-Rest of Europe-Tuberculosis
8.6.3.26 Molecular Diagnostics-Rest of Europe-Prostate Cancer
8.6.3.27 Molecular Diagnostics-Rest of Europe-Breast Cancer
8.6.3.28 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
8.6.3.29 Molecular Diagnostics-Rest of Europe-Chlamydia trachomatis/ Neisseria gonorrhoeae
8.6.3.30 Molecular Diagnostics-Rest of Europe-Tuberculosis
8.6.3.31 Molecular Diagnostics-Rest of Europe-Prostate Cancer
8.6.3.32 Molecular Diagnostics-Rest of Europe-Breast Cancer
8.6.3.33 Molecular Diagnostics-Rest of Europe-Colorectal Cancer
8.6.4 Molecular Diagnostics-Rest of Europe, by Products
8.6.4.1 Molecular Diagnostics Instruments-Rest of Europe
8.6.4.2 Molecular Diagnostics Reagents-Rest of Europe
8.6.4.3 Molecular Diagnostics Services-Rest of Europe
9 Molecular Diagnostics-Europe, by Companies
9.1 Competitive landscape
9.2 Split by Geography
9.5 Molecular Diagnostics-Spain by Companies
9.1 Molecular Diagnostics-United Kingdom by Companies
9.1 Molecular Diagnostics-France by Companies
9.1 Molecular Diagnostics-Germany by Companies
9.1 Molecular Diagnostics-Italy by Companies
9.1 Molecular Diagnostics-Rest of Europe by Companies
9.3 Molecular Diagnostics-Novartis AG-Europe
9.4 Molecular Diagnostics-Novartis AG-Europe
9.5 Molecular Diagnostics-Novartis AG-Europe
9.6 Molecular Diagnostics-Abbott Diagnostics-Europe
9.7 Molecular Diagnostics-Other Companies-Europe
9.8 Molecular Diagnostics-Abbott Diagnostics-Europe
9.9 Molecular Diagnostics-Other Companies-Europe
9.10 Molecular Diagnostics-Abbott Diagnostics-Europe
9.11 Molecular Diagnostics-Other Companies-Europe
9.12 Molecular Diagnostics-Gen-Probe Incorporated-Europe
9.13 Molecular Diagnostics-Europe-Becton Dickinson & Company
9.14 Molecular Diagnostics-Europe-Cepheid
9.15 Molecular Diagnostics-Europe-BioMérieux
9.16 Molecular Diagnostics-Europe-Siemens AG Healthcare
Please fill in the form below to receive a free copy of the Summary of this Report
Please visit http://www.micromarketmonitor.com/custom-research-services.html to specify your custom Research Requirement
| PRODUCT TITLE | PUBLISHED | |
|---|---|---|
 |
North America In Vitro Diagnostics In 2013, the Americas had the largest market, a 42% share of the Global IVD market, followed by Europe with a 31% share. However, the BRIC countries represent the fastest-growing markets due to the economic growth, the rising number of chronic diseases, and an increasing awareness about the use of IVD tests to control the spread of diseases. Moreover, the economic slowdown, pricing pressures, and high competition in mature countries will compel companies to focus on emerging markets. The report “North American In Vitro Diagnostics Market forecast for 2018 “analyzes the market by 5 segments such as reagents, instruments, techniques, applications and end-users. |
Upcoming |
 |
Europe In Vitro Diagnostics In 2013, the Americas had the largest market, a 42% share of the European IVD market, followed by Europe with a 31% share. However, the BRIC countries represent the fastest-growing markets due to the economic growth, the rising number of chronic diseases, and an increasing awareness about the use of IVD tests to control the spread of diseases. Moreover, the economic slowdown, pricing pressures, and high competition in mature countries will compel companies to focus on emerging markets. The report “European In Vitro Diagnostics Market forecast for 2018 “analyzes the market by 5 segments such as reagents, instruments, techniques, applications and end-users. Germany commanded the largest share in European IVD market. |
Upcoming |
 |
Asia In Vitro Diagnostics Asian IVD market is growing in double digit and will continue to grow in future. China commanded the largest share in Asian IVD market. The report “Asian In Vitro Diagnostics Market forecast for 2018 “analyzes the market by 5 segments such as reagents, instruments, techniques, applications and end-users. |
Upcoming |













